They have one valence electron. Noble gases have 8 electrons not one If they would have 1 electron they would be extremely reactive.
Why Are Noble Gases Very Unreactive Quora
Why are noble gases unreactive.
. They do not react with any molecule or each other. Why are noble gases called noble gases. Noble gas is non-reactive.
Neon - Ne 1s2 2s2 2p6 Argon - Ar 1s2 2s2 2p6 3s2 3p6 Krypton - Kr Ar 3d10 4s2 4p6. Elements react with one another to gain a full complement of electrons like the noble gases. Which best explains why the noble gases are chemically unreactive.
Noble gases are the least reactive of all elements. The answer to the question as to why are noble gases unreactive lies with understanding of the atomic structures of these gaseous elements. At low pressure Noble gas will conduct electricity and fluoresce.
Noble gas atoms are located in column 18 of the periodic table. They are very unreactive gases they are colourless they exist as single atoms they are monatomic The atoms of the noble gases have full outer shells of. During a chemical reaction some of the mass of the reactants is converted to energy.
According to the current theory of the atomic structure each element has a central nucleus and the electrons within it circle the nucleus continuously in an orbit. All of the noble gases except for helium contain eight electrons. Noble gases are unreactive because they have the full complement of electrons in their outermost energy levels.
124 understand why the noble gases Group 0 do not readily react. The Chemical Properties Noble gases are odorless colorless nonflammable and monotonic gases that have low chemical reactivity. Because they are already in the most stable electronic configuration they do not easily gain or lose electrons.
This is the most stable arrangement of electrons so noble gases rarely react with other elements and form compounds. The full valence electron shells of these atoms make noble gases extremely stable and unlikely to form chemical bonds because they have little tendency to gain or lose electrons. Noble gases are inert meaning that they are chemically unreactive.
With the outer s and p orbitals filled and the rule of octet satisfied the Noble Gas elements do not seek to gain or lose. Which best explains why nuclear reactions release more energy than chemical reactions. All the elements have complete electron distribution in the outer shell oxidation number 0 All the element contains High ionization energies They have Very low electronegativities.
Because theyre reluctant to share electrons from their filled outer electron shells noble gases are generally considered unreactive. Thats because they have eight valence electrons which fill their outer energy level. Explaining the inertness of noble gases When elements react their atoms complete their outer shells by.
What do all group 2 elements have in common. This group has been referred to as the inert gases indicating that they are chemically inert or unreactive. Eight electrons Why are the noble gases the most unreactive elements on the periodic table.
This is the right answer noble gases have 8 electrons that is why they are unreactive. The remaining elements in the group have full outer shells consisting of two s electrons and six p electrons for an outer shell content of eight electrons. 147 explain why substances with a simple molecular structures are gases or.
So both elements have no space or room for any more electrons so both are unreactive. It has a full outer shell and cannot incorporate any more electrons into the valence shell. Because the outer shell of valence electrons is full.
The name comes from the fact that these elements are virtually unreactive towards other elements or compounds. Noble gases have the maximum number of valence electrons and are unreactive. What do elements in the first two columns of the periodic table have in common.
Protons have a much higher mass than either neutrons or electrons. 123 Understand why elements in the same group of the Periodic Table have similar chemical properties. Neon has two energy levels.
Atoms are stable when they gain or lose electrons to have 8 electrons. The noble gases are unreactive because of their electron configurations. They are known as noble gases.
But from about 50 years noble gases are not considered as unreactive excepting helium and neon. The outer energy level or valent level is also full. They have very stable outer electron shells.
Noble gases are unreactive due to their full valence shells. Why does neon and argon not form compounds. They have eight valence electrons.
How many valence electrons do noble gases have. Group 8A The Noble or Inert Gases. Group 8A or VIIIA of the periodic table are the noble gases or inert gases.
Use the concepts of shielding and the effective nuclear charge to explain why the noble gases tend to neither give up electrons nor accept additional electrons. The Noble Gas elements are stable and unreactive inert because the outer valence shells are complete with eight electrons s2 p6. They tend to lose electrons to gain stability.
This full valence shell gives these atoms the most stable configuration they can achieve. Compared to other elements the noble gases are inert - they are extremely unreactive. This is because the noble gases valence shells are full of electrons impeding upon their ability to bond and react with other.
Helium has only one energy level which has a full valency or is full ie has no room for another electron. As a group the noble gases are very stable chemically As a group the noble gases are very stable chemically only Kr and Xe are known to form compounds. The noble gas neon has the electron configuration of 1 s 2 2 s 2 2 p 6.
They tend to form ionic bonds by losing electrons. This particular arrangement renders the atoms fairly unreactive. The noble gases are inert unreactive because they have a full outer shell of electrons.
The noble gases in order of their density are. Helium He neon Ne argon Ar krypton Kr xenon Xe and radon Rn.
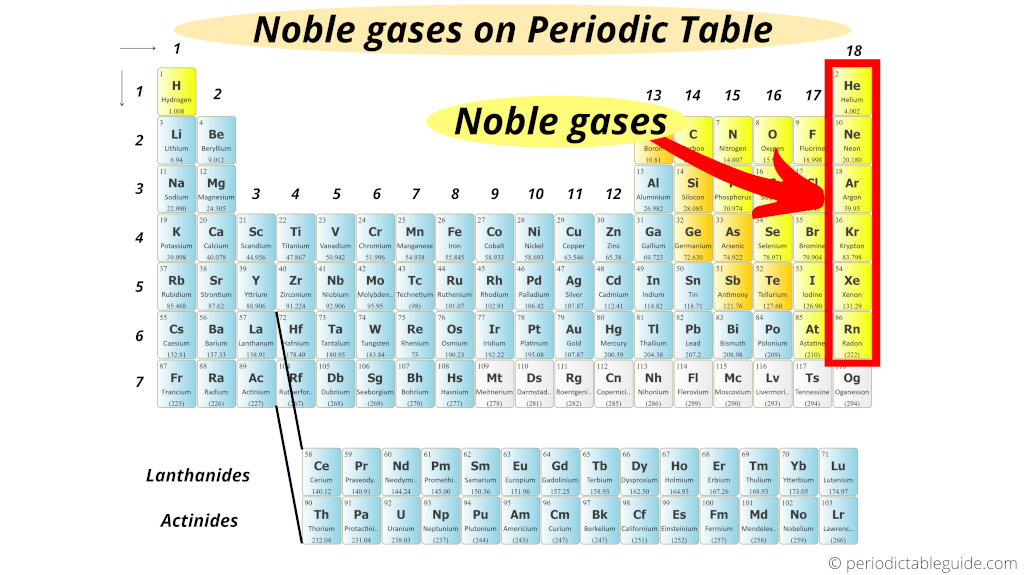
Where Are Noble Gases Located On The Periodic Table

Noble Gases Are Mostly Inert Assign Reasons
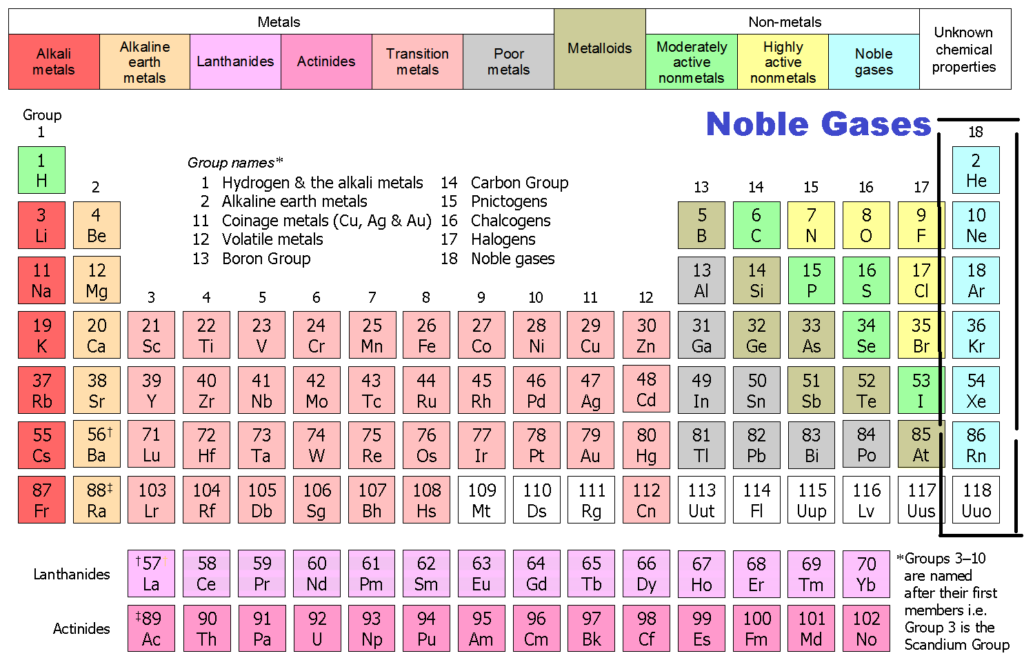
Noble Gases Why Noble Gases Are Not Reactive Configuration By Chemistry Page Medium
0 Comments